Synapses create circuits that give rise to behavior


The Higley Lab investigates the complex interplay between synaptic signaling, circuit dynamics, and behavior
using an array of methodological approaches, including electrophysiology, multiphoton and widefield imaging, optogenetics, and molecular profiling.
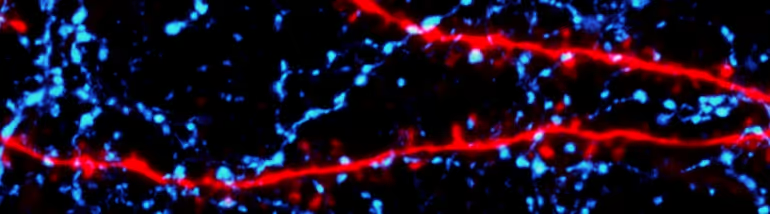
In the mammalian neocortex, neural activity is determined through an intricate dance between excitatory and inhibitory cells that are defined by an array of molecular markers, morphologies, electrical properties, and functional relationships to behavior. These cells are connected by synapses to form local and long-range circuits that organize and support cortical function. Cortical activity is also highly influenced by neuromodulatory inputs that sculpt excitability and synaptic transmission, allowing network dynamics to vary fluidly with behavioral state.
We have a long-standing interest in exploring the structure, function, modulation, and plasticity of synapses in the neocortex. Some of our work is carried out in acute brain slice preparations, giving us extremely high-resolution access to activity at individual synapses. Combining whole-cell electrophysiology with multiphoton imaging of single dendritic spines and optogenetic stimulation of targeted GABAergic interneurons, we are investigating the control of synaptic and dendritic calcium signals by inhibition. By performing axonal imaging of single presynaptic boutons, we are gaining insight into the diversity of molecular mechanisms that drive circuit-specific release of glutamate. We are also exploring how modulators like acetylcholine, norepinephrine, and endocannabinoids influence synaptic transmission, plasticity, and circuit function.
Our lab is also pioneering efforts to study synaptic transmission in the behaving animal. Applying 2-photon imaging of calcium and glutamate release with single-bouton resolution, we are linking canonical motifs of synaptic activity, such as use-dependent release probability, short- and long-term plasticity, and neuromodulation, to circuit dynamics and behavior.
We are also applying these approaches to understanding how synaptic dysfunction in disease contributes to impaired cognition. In particular, we are interested in how perturbations of synaptic function and neuromodulatory signaling contribute to non-motor symptoms and cognitive decline in Parkinson's Disease.

Most mammals use their sense of vision as a critical element of behavior, allowing them to distinguish predators and prey, find food, avoid danger, and navigate their surroundings. Visual information is encoded by neural activity along a hierarchy of circuits within the forebrain, beginning with the primary visual cortex whose diverse anatomical projections route sensory signals to various downstream locations that serve different aspects of perception, learning, and motor output.
We are actively exploring how animals use visual information to navigate the world and the cortical circuits that support knowledge and memory of their location relative to behaviorally relevant objects. We are applying widefield and multiphoton imaging to monitor neural activity in areas like visual, retrosplenial, and entorhinal cortex. These studies are carried out in both head-fixed mice navigating virtual reality environments and freely moving animals. Imaging allows us to follow the same neurons over weeks, identifying cell types by genetic expression of indicators and linking their activity to sensory cues, location, and task performance. We are also using Neuropixels to obtain dense, high-temporal-resolution electrophysiological recordings simultaneously across visual areas, providing novel insights into the patterns of neural activity during goal-directed behavior.
These experimental studies are combined with novel computational strategies to discover the dynamic organization and geometry of neural networks associated with task learning and performance. We use both traditional linear models and machine learning-based strategies to understand how cellular and circuit activities support flexible behavior.

The use of light to both monitor and manipulate cellular activity is continuing to transform the study of biological systems, including the brain. In particular, fluorescence imaging enables us to study the structure and activity of synapses, cells, and circuits, spanning several orders of spatial magnitude. Our lab uses optical tools, including widefield and multiphoton imaging, optogenetic activation and suppression, and light-based tagging of targeted cell populations, to investigate the causal relationships between neural signaling and behavior. With these tools, we are observing glutamate release at single synapses across spontaneous fluctuations in arousal, neuronal spiking aligned to task performance, and patterned network activity associated with learning-dependent neural plasticity.
We are also developing new light-based tools and strategies for neuroscience applications. We described a combination of widefield, "mesoscopic" imaging with 2-photon microscopy to provide simultaneous recordings of individual neurons and cortex-wide networks and reveal large-scale functional connectivity of genetically identified cell types. In addition, we are combining adaptive optics and 3-photon imaging to monitor activity in deep brain regions like the entorhinal cortex of mice navigating virtual environments.
Most advanced imaging techniques have required the animal to be head-fixed under the optics of a complex microscope, and this substantially restricts the range of behaviors that can be studied. To address this challenge, we have been developing a new system, the Miniature Mesoscope or "MiniMe", that enables non-invasive, widefield fluorescent imaging of the entire dorsal cortex of freely moving mice. We are using this system to examine the flexibility of network dynamics under conditions of changing behavioral contingencies. In addition, we are working to make this extremely cost-efficient strategy widely available to the broader community.
We are grateful to the following funders for supporting our work!









